.png)
.png)
We’ve fundamentally redesigned water electrolysis to create the ideal platform
for producing Green Hydrogen
We’ve fundamentally redesigned water electrolysis to create the ideal platform for producing Green Hydrogen
We envision a solution rooted in launching the green hydrogen era, enabling
low-carbon green energy and hydrogen fuels to be supplied to sectors in future cities
at a competitive price at scale.
We envision a solution rooted in launching the green hydrogen era, enabling low-carbon green energy and hydrogen fuels to be supplied to sectors in future cities at a competitive price at scale.
With the goal of rendering carbon-intensive industry a thing of the past,
we have developed cost-competitive technologies
for the production of green hydrogen.
With the goal of rendering carbon-intensive industry a thing of the past, we have developed cost-competitive technologies for the production of green hydrogen.
Hydrogen's abundance and unique properties make it a key piece in the puzzle of our world's energy solutions.It is an excellent source of renewable energy ideally suited to power buses, trucks, and other heavy vehicles and it can produced and used cleanly. Hydrogen is a store of energy, playing the role of a battery. Hydrogen can be produced when the natural resource is available and stored for when it is needed. It can also support the electricity grid, providing demand control during peak times, and enabling more efficient use of our electrical infrastructure. Hydrogen can redefine energy future
Hydrogen's abundance and unique properties
make it a key piece in the puzzle of our
world's energy solutions.It is an excellent source
of renewable energy ideally suited to
power buses, trucks, and other heavy vehicles
and it can produced and used cleanly.
Hydrogen is a store of energy, playing
the role of a battery. Hydrogen can be produced
when the natural resource is available and
stored for when it is needed. It can also support
the electricity grid, providing demand control
during peak times, and enabling more
efficient use of our electrical infrastructure.
Hydrogen can redefine energy future

Zero Emissions
When using hydrogen
for power or heat it
gives off water vapour
with no carbon
emissions.

Clean
Hydrogen has no
colour or odour and is
non-toxic and
environmentally
benign.
Diffuses Quickly
Being 14 times
lighter than air,
when outside
it's container, it
dissipates quickly.

Plentiful
Hydrogen is the most
abundant element in
the universe and every
region in the world can
create it.

Energy Rich
One kilogram of
hydrogen holds three
times the energy of
one kilogram of crude
oil.

Life-Giving
The sun is powered
by hydrogen and
it's in our water
so it is the basis
of life.
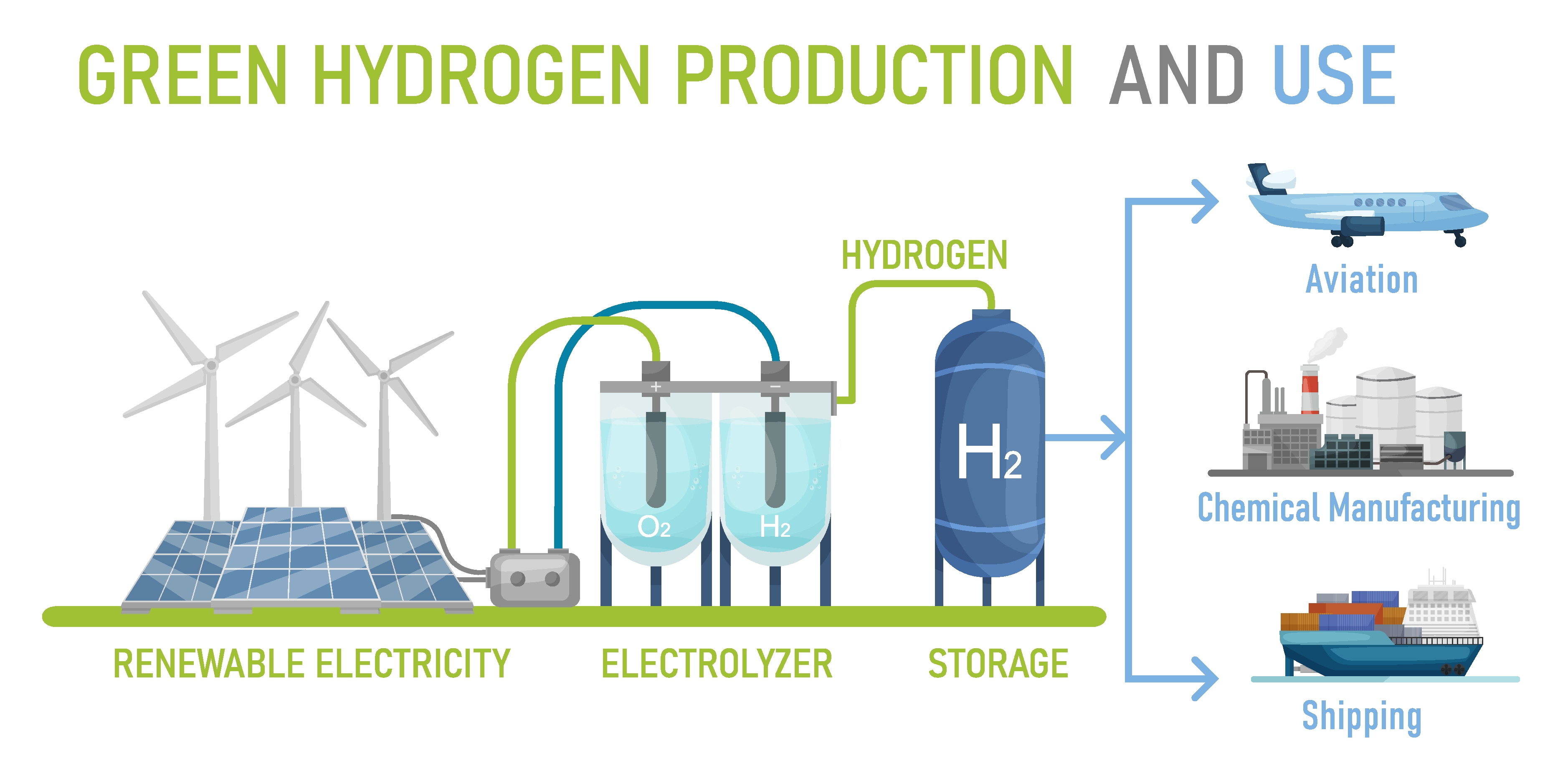
Green hydrogen is being produced by electrolysis, which is a process that uses electricity to split water molecules (H2O) into hydrogen (H2) and oxygen (O2). When electrolysis is combined with renewable electricity, the production of fuels and chemicals can be decoupled from fossil resources, paving the way for an energy system based on 100% renewable energy. Electrolytic hydrogen production faces technological challenges to improve its efficiency, economic value and potential for global integration. In conventional water electrolysis, the water oxidation and reduction reactions are coupled in both time and space, as they occur simultaneously at an anode and a cathode in the same cell. This introduces challenges, such as product separation, and sets strict constraints on material selection and process conditions. Here, we decouple these reactions by dividing the process into two steps: an electrochemical step that reduces water at the cathode and oxidizes the anode, followed by a spontaneous chemical step that is driven faster at higher temperature, which reduces the anode back to its initial state by oxidizing water. This enables overall water splitting at average cell voltages of 1.44–1.60 V with nominal current densities of 10–200 mA cm−2 in a membrane-free, two-electrode cell. This allows us to produce hydrogen at low voltages in a simple, cyclic process with high efficiency, robustness, safety and scale-up potential. In our process we produce hydrogen and oxygen in two separate steps in the first step we produce hydrogen at the cathode while the anode is being charged without producing oxygen, this step is carried out at room temperature then in the second step we heat up the anode to discharge it without electricity in a completely chemical and spontaneous process during this process the anode releases the oxygen once it's discharged we can place it back in the cold solution to continue hydrogen production.
Green hydrogen is being produced by electrolysis,
which is a process that uses electricity to split
water molecules (H2O) into hydrogen (H2) and oxygen (O2).
When electrolysis is combined with renewable electricity,
the production of fuels and chemicals can be decoupled
from fossil resources, paving the way for an energy system
based on 100% renewable energy.
Electrolytic hydrogen production faces technological
challenges to improve its efficiency, economic value and
potential for global integration. In conventional water
electrolysis, the water oxidation and reduction reactions
are coupled in both time and space, as they occur simultaneously
at an anode and a cathode in the same cell. This introduces
challenges, such as product separation, and sets strict
constraints on material selection and process conditions. Here,
we decouple these reactions by dividing the process into two steps:
an electrochemical step that reduces water at the cathode and
oxidizes the anode, followed by a spontaneous chemical step
that is driven faster at higher temperature, which reduces
the anode back to its initial state by oxidizing water. This enables
overall water splitting at average cell voltages
of 1.44–1.60 V with nominal current densities of 10–200 mA cm−2
in a membrane-free, two-electrode cell. This allows us to produce
hydrogen at low voltages in a simple, cyclic process with
high efficiency, robustness, safety and scale-up potential.
In our process we produce hydrogen and oxygen in two separate
steps in the first step we produce hydrogen at the cathode while
the anode is being charged without producing oxygen, this step is
carried out at room temperature then in the second step we heat up
the anode to discharge it without electricity in a completely
chemical and spontaneous process during this process the anode
releases the oxygen once it's discharged we can place it back in
the cold solution to continue hydrogen production.
.png)
Electrolysis requires only the cathode, the anode,
and the electrolyte of nickel hydroxide,
and is achieved by this equation:
4H2O + 4e– → 4OH– + 2H2
OER: 4OH– → 2H2O + O2 + 4e–
(Ni(OH)2 + OH– → NiOOH + H2O + e– ) × 4
HER: 4H2O + 4e– → 4OH– + 2H2 25 °
4NiOOH + 2H2O → 4Ni(OH)2 + O2
Electrolysis requires only the cathode, the anode, and the electrolyte of nickel hydroxide, and is achieved by this equation:
4H2O + 4e– → 4OH– + 2H2 OER: 4OH– → 2H2O + O2 + 4e– (Ni(OH)2 + OH– → NiOOH + H2O + e– ) × 4 HER: 4H2O + 4e– → 4OH– + 2H2 25 ° 4NiOOH + 2H2O → 4Ni(OH)2 + O2

Our first product will be a 500 kg/day electrolyzer, operating at 50 bar, It will be delivered in a container form factor. Our first commercial systems will consume about 42 kW/h per kg of hydrogen produced.
Our first product will be a 500 kg/day electrolyzer, operating at 50 bar,
It will be delivered in a container form factor.
Our first commercial systems will consume about 42 kW/h
per kg of hydrogen produced.
.png)

